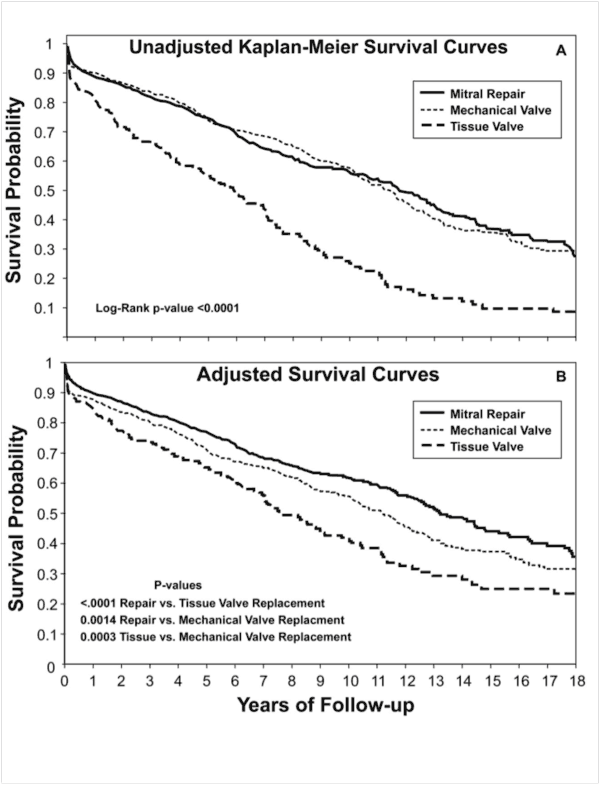
Figure 1
Legend: Survival after isolated mitral procedures at Duke University 1986-2006 (n=2064). Panel A: Unadjusted raw survival data for valve repair, mechanical valve replacement, and tissue valve replacement. Panel B: Risk-adjusted survival using a Cox proportional hazards model to account for differences in baseline characteristics between groups and also in propensity to choose various procedures. Mitral valve repair achieved the best outcomes and tissue valve replacement the worst.

Figure 2
Legend: Sequential steps in “adjustable artificial chordal replacement. A: A ruptured chord from the posterior papillary muscle to the posterior leaflet is evident. B: A pledgetted anchor suture is placed longitudinally in the papillary muscle, and a 2-0 Gore-Tex artificial chord is passed through the anchor pledget and left untied. C: After Carpentier ring placement (AnnuloFlow, Sorin Group), the chord is retrieved and woven as a loop into the leaflet in 3 full-thickness bites: free edge, flaring laterally through the surface of coaptation, and then angling back medially through the line of coaptation, emerging onto the atrial surface of the leaflet. D: The chordal slip knot is adjusted during valve testing to produce good anterior-posterior and left to right leaflet symmetry, and no residual leak. The knot is then tied tightly and completed.
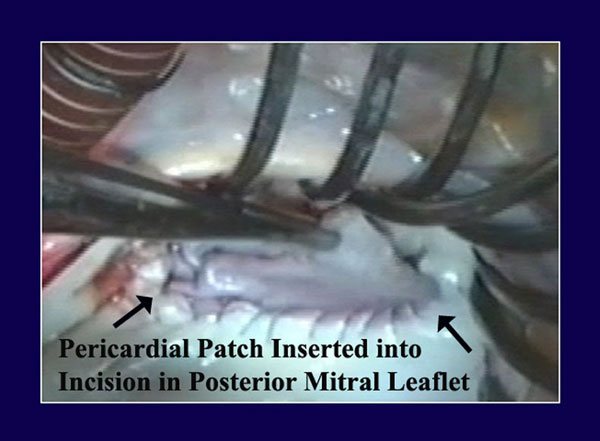
Figure 3
Legend: Autologous pericardium has been lightly tanned by a 10-minute immersion in 0.6% gluteraldehyde, cut to size, and sutured to the annular aspect of an incision in the posterior mitral leaflet. The top part of the patch will then be sutured to the leaflet side of the incision, completing pericardial patch augmentation of the posterior leaflet. This is useful with rheumatic scarred retracted leaflets, but also with ischemic leaflet tethering or inadequate leaflet tissue associated with pure annular dilatation.
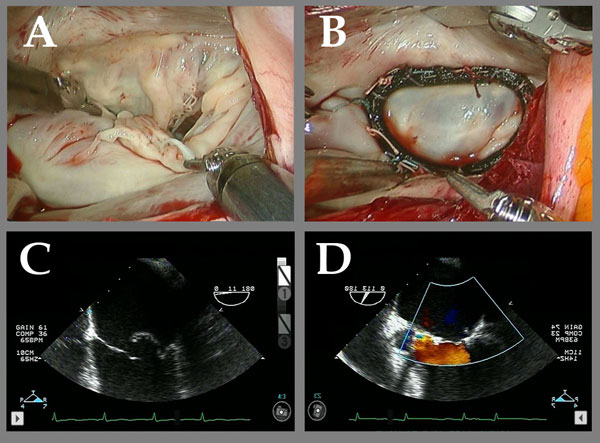
Figure 4
Legend: Robotic ACR of a valve with multiple posterior leaflet chordal ruptures. A and C: The flail posterior leaflet is evident. B: After placement of a single ACR from the posterior papillary muscle to the posterior leaflet, the valve is completely competent. D: After ACR, the prolapsing posterior leaflet is well below the annular plane, with a good surface area of coaptation, and complete recovery of competence. Chordal length adjustment was performed after placement of the full annuloplasty ring (Memo 3D, Sorin group).
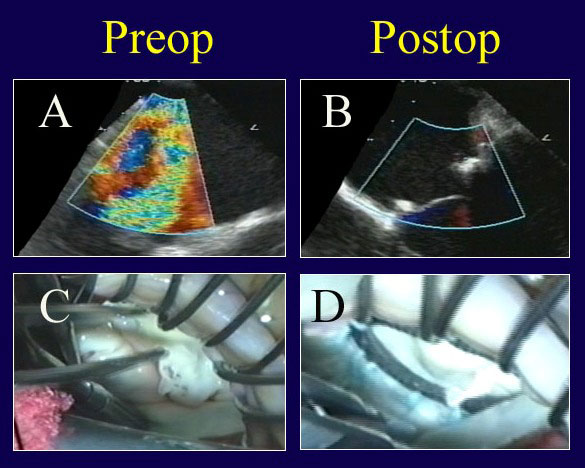
Figure 5
Legend: Echo and video findings in a patient with true commissural prolapse, before and after ACR repair. A: Severe MR is evident, and components of anterior, commissural, and posterior leaflet prolapse were visualized. B: After ACR repair, the valve is completely competent with good leaflet position. C: A “windsock” of true commissural prolapse is present. D: Valve symmetry and competence are normal after ACR placement to both leaflets and full ring annuloplasty (AnnuloFlow, Sorin Group).
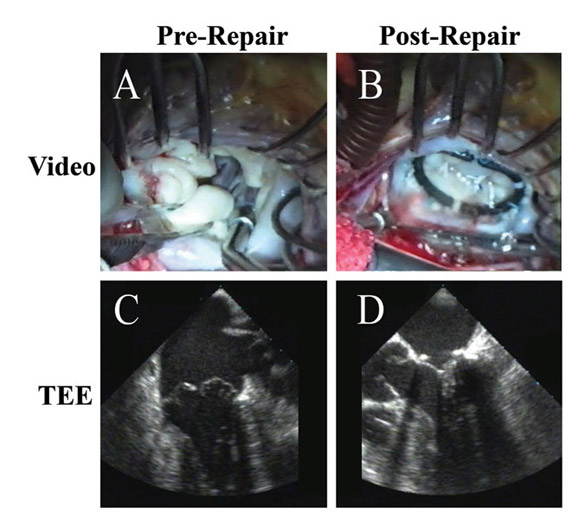
Figure 6
Legend: ACR repair of a Barlow’s valve with multi-segmentation, generalized prolapse, and annular calcification. A: The repair is begun by limited decalcification of the annulus (enough to allow forward annular positioning with ring placement). Multiple prolapsing segments are evident. B: After ring annuloplasty (AnnuloFlow, Sorin Group) and placement of 4 sets of artificial chords (with length adjustment of 2), the valve is fully competent. C: Typical generalized prolapse associated with a Barlow’s valve is evident on echo, also with a severe central jet of regurgitation. D: After ACR repair, leaflet position is excellent with a good surface area of coaptation and no residual leak. Leaflet is never resected, and the authors believe that the concept of “excess leaflet tissue” is not applicable to this situation – the more leaflet surface area, the better the competence.
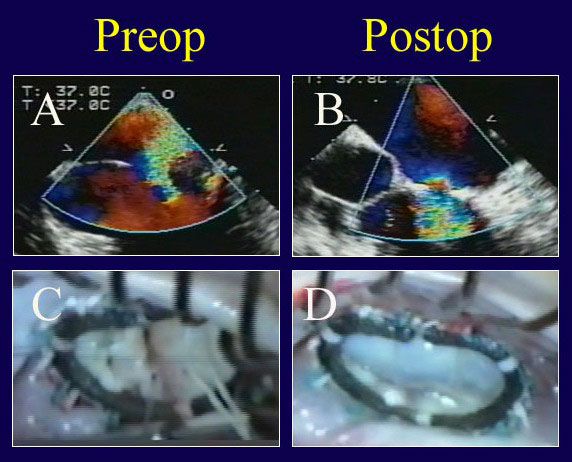
Figure 7
Legend: ACR repair of a patient with fully treated endocarditis. A: The echo exhibits severe valve leak. B: After repair with suture closure of multiple posterior leaflet holes and ACR to both leaflets, the valve is fully competent. C: The multiple holes in the posterior leaflet are evident; these are sutured closed and ACR’s are being performed after ring placement (AnnuloFlow, Sorin Group). D: With valve testing after ACR repair, the symmetry of the competent valve appears normal.
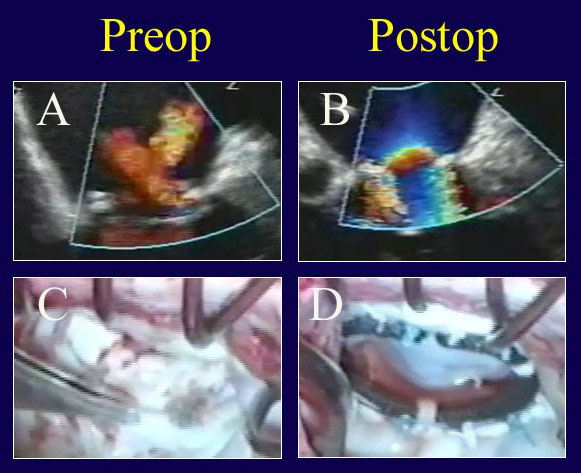
Figure 8
Legend: Re-repair of a prolapse valve after failed reconstruction. A: Two jets of regurgitation are evident – one from a leak through a suture hole in the anterior aspect of the ring sutures, and a second from an inadequate posterior leaflet after over-resection. The leaflet hole is closed, an Alfieri stitch is taken down, a posterior leaflet autologous pericardial patch is placed to compensate for insufficient posterior leaflet surface area (C), and a Gore-Tex ACR is placed to a prolapsing commissural cusp (that was overlooked at the first procedure). After ring placement (true-sized to a smaller diameter; AnnuloFlow, Sorin Group), the chord is adjusted once, and the valve is fully competent (B). The tied ACR and the good leaflet symmetry are shown in panel D.
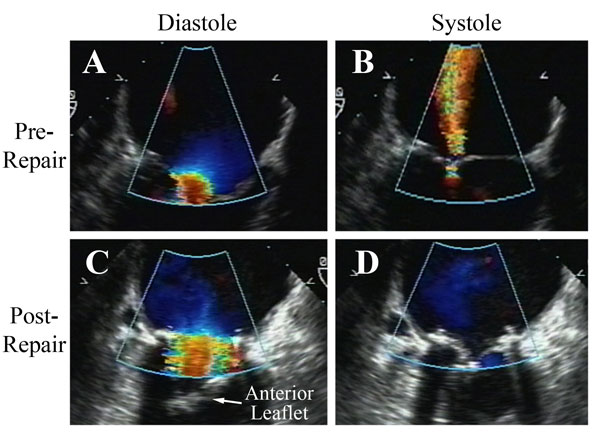
Figure 9
Legend: A patient with long-standing rheumatic mitral stenosis and regurgitation undergoing a rheumatic repair. A and B: On pre-bypass echo, the stenotic and insufficient mitral valve is demonstrated. C and D: After insertion of an autologous pericardial patch into the posterior leaflet, decalcifying commissurotomy, resection of the submitral apparatus to the anterior leaflet, and re-attachment of the mobile anterior leaflet to both papillary muscles with adjustable ACR, the valve is fully competent with insignificant gradient. A full rigid ring also is placed (AnnuloFlow, Sorin Group). These types of more comprehensive ACR repairs for rheumatic valves have been quite stable in the intermediate-term.
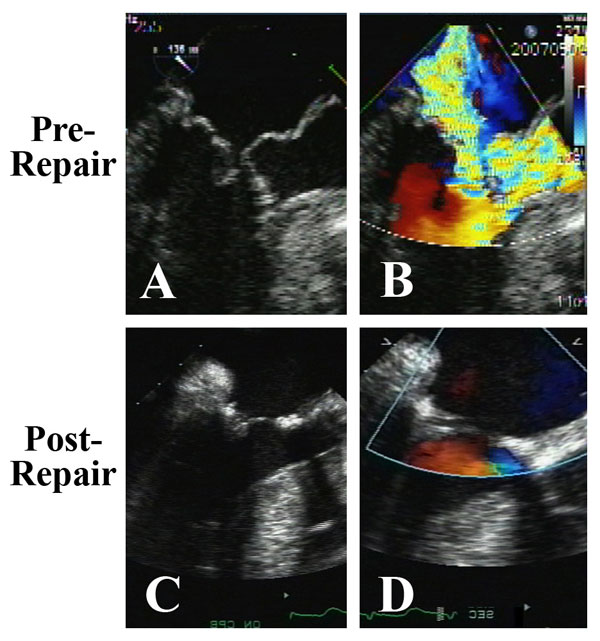
Figure 10
Legend: ACR mitral repair in a patient with HOCM crisis and severe mitral anomalies. A and B: Pronounced systolic anterior motion of the anterior mitral leaflet and anterior papillary muscle prolapse against an asymmetrically hypertrophied septum produces severe outflow tract obstruction and mitral regurgitation. C and D: After septal myectomy, resection of the obstructing anomalous anterior papillary muscle, and mitral reconstruction with ACR and ring annuloplasty (AnnuloFlow, Sorin Group), the outflow obstruction is relieved, the systolic anterior motion is eliminated, and the valve is fully competent. This type of repair has been quite stable in the intermediate-term.
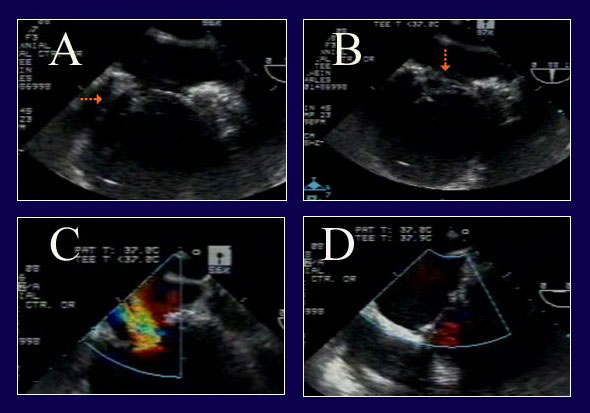
Figure 11
Legend: ACR repair of complex IMR in a re-operative patient with severe LV dysfunction. A: The posterior leaflet does not close properly due to tethering (arrow) from posterior wall infarct expansion. B: However, on a different view, a segment of anterior leaflet also is prolapsing (arrow) due to papillary muscle elongation. C: The severe MR from both leaflet abnormalities is evident. D: After pericardial patch augmentation of the posterior leaflet and ACR to the anterior leaflet, the valve is fully competent. Full ring annuloplasty also was performed (AnnuloFlow, Sorin Group).
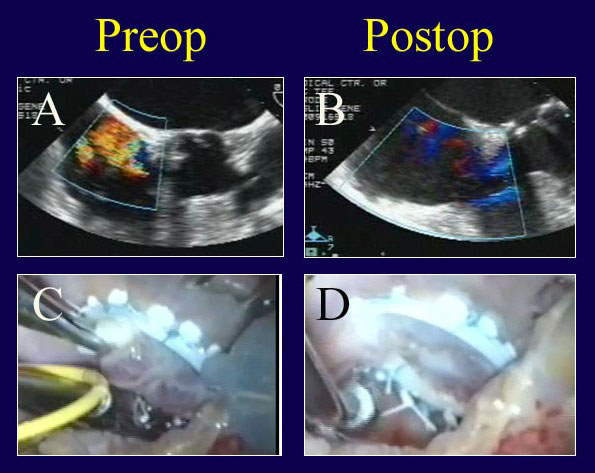
Figure 12
Legend: A patient undergoing mitral and tricuspid valve repair is thought to have severe TR for functional reasons (A). However, after Carpentier tricuspid ring annuloplasty (Edwards Lifesciences), a prominent prolapse of the anterior tricuspid leaflet and persistent valve leak are noted (C). ACR is performed from an RV papillary muscle to the anterior leaflet (D) with complete recovery of competence (B).

Figure 13
Legend: Mitral repair rates and operative mortalities for all mitral procedures (including multiple valves) in the author’s practice (27). The recent increase in repair rates toward 100% has been due to perfection of ACR techniques, together with aggressive use of pericardial patches and full rings. With increasing repair (and certainly for other reasons) operative mortality now is near-zero. These data support the increasing use of valve repair across the spectrum of mitral pathologies.
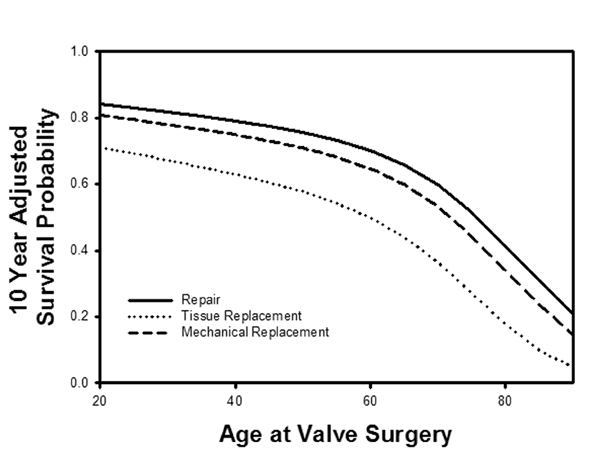
Figure 14
Legend: A: Long-term survival characteristics after mitral procedures for all pathologies at Duke University over a 20-year period. Mitral repair was associated with a significantly better risk-adjusted survival than valve replacement, and tissue valve replacement was the worst. Risk-adjusted 10-year survival was better with mitral repair for all patient ages at the time of implant. At no patient age did tissue valve replacement achieve equivalent outcomes to either mechanical valves or repair, but repair was clinically and statistically the best (30)
prada birkenstock ropa prada hombre openai chatgpt plus amazing grace in cherokee native american instagram followers ads vinyl building material track followers instagram long chin beard digital offset press chatgpt wordpress plugin dr kiran patel house brown prada shoes uggs usa outlet online followers pro instagram wearing prada prada cardholder prada mens gloves prada dog clothes gucci bag outlet usa prada luna sport komori digital press bing chatgpt download factory stores online chatgpt for windows prada blue bags prada paintings bonnie lure team concepts printing analyze instagram followers www.gucci.com usa prada brasil simon premium outlets vip non followers instagram wiki chatgpt chatgpt for seo prada panier bag prada law prada boots white prada bottle prada leather prada sport glasses prada official website luxe store prada vogue runway gucci online shopping shannon lutz instagram block followers prada shoulder bags siding estimate worksheet birkenstock outlet store mosaic company florida locations bing chatgpt download prada leopard bag chatgpt plus price big cork boards remove instagram followers instagram followers analysis prada sizing tamagotchi bulletin board walls prada glasses. large bulletin board prada denim bra prada sport sandals prada necktie indigo pantone color chatgpt model size captain epaulets carlucci s diabo veste prada prada nylon strap lars make up gucci things gucci outlet bags prada shies cartera prada negra jude and i construction copilot vs chatgpt silver prada vintage prada dress colored cork boards cost of chatgpt blue prada glasses chatgpt release englewood ymca prada sneaker green kiran c patel silicone head luxury designer outlet instagram followers ads prada instagram elements construction large prada backpack rambutan ripe zhou feng chen prada eyeglasses case private instagram followers outlet sale online vintage prada loafers boss outlet store prada illusion sunglasses get chatgpt tamagotchies openapi chatgpt brunello cucinelli outlet online mosaic tampa fl
